
Batteries must match growing demands from greater electrical loads
By David Osborne

AC Transit, the third largest public bus system in the state of California, serving the Alameda-Contra Costa Transit District in the Bay Area, launched the HyRoad program in 1999 to demonstrate the viability of zero-emission public transit. In conjunction with bus manufacturer Van Hool, the agency introduced a fleet of three hydrogen fuel cell buses that operated for nearly 225,000 miles with an overall fuel economy of 6.60 miles per kg., which equates to 7.55 miles per diesel equivalent gallon. During the same period AC Transit’s diesel buses had a fuel economy of 4.2 mpg, which equated to the fuel cell buses having a 61 percent better fuel economy than the diesel buses. The initial demonstration project was a tremendous success.
Fast forward to 2009 when Van Hool and AC Transit started looking for a new partner to provide an energy storage system for the next generation of fuel cell buses. The goal was to manufacture a total of 12 fuel cell buses using new technologies to increase efficiencies and performance in providing service to cities around the Bay Area. AC Transit now operates the largest single fleet of fuel cell buses in the U.S.
EnerDel, Indianapolis, IN, provided its lithium-ion energy storage solution to capture the energy created by the fuel cell as part of an integrated energy propulsion system.
Lithium-ion cells, the core component of a lithium-ion battery, use a cathode (positive electrode) and an anode (negative electrode) coupled with an electrolyte to serve as a conductor between the two electrodes. The cathode uses a mixed oxide and the anode consists of hard carbon. During charge and discharge, the ions flow from the anode to the cathode to create, store and release the energy.
To meet the desired energy and capacity required for the application, a specific number of battery pack modules containing a specific number of lithium-ion cells insert into the finished battery pack. The AC Transit hybrid-electric fuel cell buses use the on-board fuel cell system to combine oxygen from the air with hydrogen from the tanks to produce water vapor and enough electricity to power the bus, which integrates with the lithium-ion battery pack and battery management system to manage the power system that propels the bus. Braking energy captured by the drive motors is also stored in the EnerDel lithium-ion batteries for extra power on demand.
Integrated energy storage
EnerDel provided its PE350-689 Vigor+ Battery Pack, which provides a rated capacity of 35Ah with an energy rating of 21.5kWh. The high charge and discharge rates of this battery pack delivers the performance and reliability required for the power system developed for the Van Hool fuel cell bus. The EnerDel battery pack is designed with proprietary prismatic cell technology that provides one of the highest energy densities in the industry (>145 Wh/kg).
EnerDel’s battery pack incorporates the T100 Assure+ Battery Management System (BMS), which integrates the battery pack with the fuel cell and continually communicates with the entire vehicle drive system with regard to efficiency and safety. The BMS also controls and manages the lithium-ion cells in the battery modules, including balancing the energy of the system at the cell level. It calculates and manages system parameters and provides diagnostics for all control system components.
To date, the AC Transit hydrogen fuel cell hybrid bus fleet has traveled more than 550,000 zero-emissions miles and carried more than 1.8 million passengers. BR
David Osborne serves as program management director for Transportation, EnerDel, Indianapolis, IN.
A word about Lithium and Lithium-ion batteries
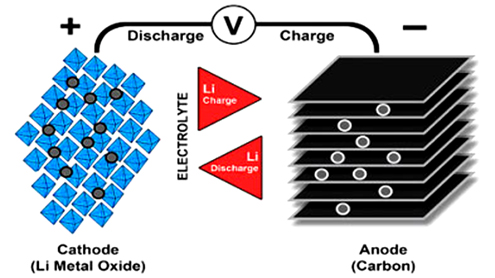
Lithium is the lightest of all metals, has the greatest electrochemical potential and provides the largest specific energy per weight. Rechargeable batteries with lithium metal on the anode (negative electrodes) could provide extraordinarily high energy densities. However, the inherent instability of lithium metal, especially during charging, shifted research to a non-metallic solution using lithium ions. Li-ion batteries come in many varieties, but all share the catchword, lithium-ion. Although strikingly similar at first glance, these batteries vary in performance, and the choice of cathode materials gives them their unique characteristics.
Although lower in specific energy than lithium-metal, Li-ion is safe, provided cell manufacturers and battery packers follow safety measures in keeping voltage and currents to secure levels. Today this chemistry has become the most promising and fastest growing on the market. Meanwhile, research continues to develop a safe metallic lithium battery.
The specific energy of Li-ion is twice that of NiCd, and the high nominal cell voltage of 3.60V as compared to 1.20V for nickel systems contributes to this gain. Improvements in the active materials of the electrode have the potential of further increases in energy density.
The load characteristics are good, and the flat discharge curve offers effective utilization of the stored energy in a desirable voltage spectrum of 3.70 to 2.80V/cell. Nickel-based batteries also have a flat discharge curve that ranges from 1.25 to 1.0V/cell. BR
